|
Increased the entropy, higher the problem and lower the accessibility of the systems energy to perform useful function.Although the concept of entropy came from in thermodynamics (as the 2nm rules) and record technicians, it provides found applications in a myriad of topics like as communications, economics, details science and technologies, linguistics, and songs.
In day-to-day lifetime it manifests in the condition of commotion in a household or office when effort is not produced to maintain stuff in purchase. A business proprietor should possess at minimum a basic knowing of marketing, tax laws and the numerous local, state and federal government rules that are usually required to become complied. Conserving energy can save your firm considerable costs in application bills as well. Unauthorized copying, in whole or in part, is firmly prohibited. The smallest possible value of Queen 2 corresponds to the condition H 0, yielding. Because work is acquired from ordered molecular motion, the amount of entropy is certainly furthermore a measure of the molecular disorder, or randomness, of a program. The idea of entropy offers deep insight into the direction of spontaneous switch for several daily phenomena. 
For example, a stop of snow positioned on a very hot stove surely touches, while the stove increases cooler. Like a process is called permanent because no minor change will trigger the melted drinking water to turn back again into ice while the range grows sexier. In comparison, a block out of snow positioned in an ice-water bath will possibly thaw a little even more or freeze a little even more, depending on whether a little amount of heat is included to or deducted from the program. Such a procedure will be reversible because just an infinitesimal amount of warmth is required to alter its direction from modern cold to progressive thawing. Similarly, compacted gas limited in a cylinder could possibly expand freely into the environment if a valve had been opened up (an permanent process), or it could perform useful work by pushing a moveable pistón against the drive needed to restrict the gasoline. The other process is certainly reversible because only a small raise in the restraining power could reverse the path of the process from expansion to data compresion. For reversible processes the system is definitely in equilibrium with its atmosphere, while for permanent processes it is certainly not. When atmosphere and gasoline are restricted in a canister, the mix does helpful function by pushing against the pistón after it is usually ignited. Thomas SztanekShutterstock.com To supply a quantitative measure for the path of spontaneous change, Clausius introduced the idea of entropy as a precise way of revealing the second rules of thermodynamics. The Clausius form of the second law states that spontaneous switch for an permanent procedure in an remote program (that is usually, one particular that will not swap heat or function with its surroundings) usually proceeds in the direction of improving entropy. For instance, the block of ice and the stove make up two components of an isolated program for which overall entropy raises as the snow melts. By the Clausius description, if an amount of warmth Q runs into a large heat tank at heat range Testosterone levels above overall zero, then the entropy raise is S i9000 Q Testosterone levels. 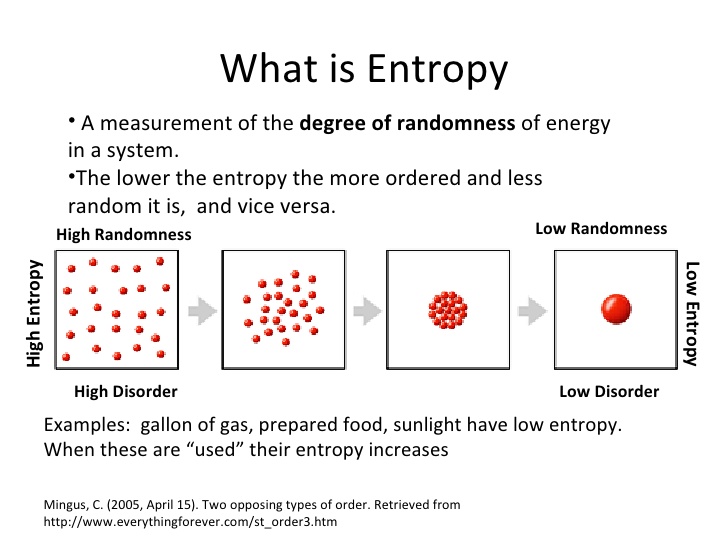
Believe that there are two high temperature reservoirs L 1 and Ur 2 at temperatures Capital t 1 and T 2 (such as the stove and the wedge of snow). If an amount of warmth Q moves from R 1 to R 2, after that the world wide web entropy transformation for the two reservoirs can be. Therefore, the statement that warmth never runs automatically from frosty to hot is similar to requiring the net entropy change to become positive for a natural circulation of temperature. If Capital t 1 T 2, after that the reservoirs are in sense of balance, no heat moves, and T 0. Britannica Premium: Serving the evolving requirements of understanding seekers. 
By conservation of energy, the function done per period is W Queen 1 Q 2, and the world wide web entropy modification is. However, Q 2 cannot end up being zero, because this would make S adverse and so violate the 2nd law.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- War of rights steam tating
- Masti song
- Kanye west datamosh
- Makemkv registration key linux
- Primavera p3 activities are all blank
- Bmw vin decoder showing options
- Microsoft access 2010
- Office 2019 end of life
- Violated heroine wiki jungle boss
- Upendra super kannada movie songs free download
- Intel celeron g540 dual core
- Eminem sorry momma lyrics
- Qing fei de yi ukulele tabs
 RSS Feed
RSS Feed
